Optical Coherence Tomography and OCT Angiography: Clinical Reference And Case Studies
Darrin A. Landry, CRA, OCT-C
Amir H. Kashani, MD, PhD
Bryson Taylor Publishing, 2016
ISBN–‐13:978–‐1523976867
ISBN–‐10:1523976861
www.BrysonTaylorPublishing.com
In the early days of retinal angiography, photographers often worked very closely with ophthalmologists, learning together as they explored the diagnostic uses of fluorescein angiography and unraveled the complexities of interpreting the fascinating images they were capturing. This spirit of scholarly collaboration between imager and physician continues today in a new book: Optical Coherence Tomography and OCT Angiography, Clinical Reference And Case Studies by Darrin Landry and Amir Kashani. These authors are both well respected in their respective fields as educators and authors. Together they have created a timely textbook that will appeal to members of both professions.
Before receiving an advance copy of this book for review, I anticipated that the content would focus almost exclusively on OCT angiography. I was pleasantly surprised to find that although the book features OCT-A prominently, it is much more than a text on this new state-of-the-art technology. It appropriately places OCT-A in the context of multiple imaging modalities to assist in diagnosis of a variety of retinal conditions.
The authors have produced a book that is part tutorial, part clinical atlas, and a collection of over forty cases that “puts it all together” using multiple imaging modalities with clinical descriptions. The book is divided into three sections:
Section 1. OCT and OCT Angiography
The introductory section will be particularly useful to imagers as it includes a basic overview of OCT and OCT-A technology, followed by a discussion on pattern recognition, normal anatomy and layers of the retina, how to move the scan pattern, recognizing artifacts, EDI/FDI and a basic primer on OCT-A. The OCT-A primer explains how this technology scans through the z-axis and detects motion to identify the retinal vasculature including the deep retinal plexus. It includes a discussion of artifacts specific to OCT-A . This section will be especially helpful to those new to OCT and OCT-A, and anyone preparing for certification as an OCT-C.
Section 2. Atlas of Images and Disease Pathology
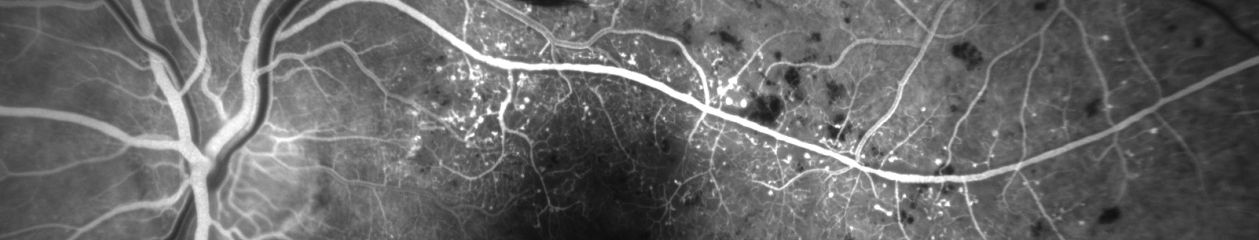
This section is an atlas of retinal OCT findings organized in anatomical order from the vitreous to the choroid. For each condition, the text includes a brief discussion of the disease process, clinical findings, and appearance in multiple modalities. For each condition, there are multiple image examples providing a full spectrum of potential findings for that disease. For instance, there are over twenty different examples of epiretinal membrane. Novice imagers will find this variety especially helpful in learning to recognize different manifestations of a single condition. In addition to common retinal findings the book also includes good examples of less recognized conditions such as outer retinal tubulation (ORT) and reticular pseudodrusen. As expected, retinal vascular diseases include OCT-A examples along with SD-OCT and other imaging modalities including fluorescein and ICG angiography. Experienced imagers will recognize many of these conditions, but the addition of OCT-A will give them another viewpoint and expand their understanding of each disease.
Section 3. Case Studies
The final section of the book is a series of over forty cases where the authors combine a brief medical summary with appropriate imaging modalities for clinical correlation. This format fits well with the current trend of “case-based-learning” in medical education. In many of these cases, OCT-A dovetails nicely with other imaging modalities to increase our understanding of a disease process or help confirm a diagnosis. This quote from the book’s Preface describes the format well “These images are presented in the context of additional imaging modalities to aide the reader in making useful correlations.”
In conclusion, this timely book is well organized and thorough, without becoming unwieldy. It is easy to navigate between sections if you want a quick reference on OCT anatomy or to look for examples of specific retinal conditions and how they may appear on OCT, OCTA and other imaging modalities. With over a thousand images and forty cases, to say that this book is generously illustrated would be an understatement. It is an impressive collaboration between an ophthalmic imager and a retinal specialist that should appeal to a wide audience that would include ophthalmic imagers, retinal technicians, residents in training, and clinicians wanting a reference for clinical correlation between modalities.
From a personal standpoint, I think it’s great to have an ophthalmic imager making a significant contribution to the ophthalmic literature. Darrin’s collaboration with Dr Kashani serves as a model for what imagers can accomplish when we collaborate with physicians on a professional level. The spirit of collaboration between professions is echoed several times in the book including this statement from the Introduction, “Constant and close communication between the physician and imager is very essential.”
Reviews like this often end with a cliché that suggests that everyone in the profession should “add this book to your collection” or “keep a copy on your bookshelf”. I’ve tried to avoid those clichés, but honestly, I am happy to have this book in my collection and plan to keep it handy in clinic for reference, especially as we integrate OCT-A into our own diagnostic armamentarium.





















 The footprint of the Eidon is fairly compact, but the instrument is taller than most fundus cameras. The device is operated via touch screen tablet and has both automatic and manual controls. The Eidon has a fixed 60 field of view, but is capable of capturing several fields and creating montage images. It features a 14 megapixel sensor to capture color, red free, and infrared images. The red free photos are extracted from the color image rather than through a separate exposure with a blue-green light source.
The footprint of the Eidon is fairly compact, but the instrument is taller than most fundus cameras. The device is operated via touch screen tablet and has both automatic and manual controls. The Eidon has a fixed 60 field of view, but is capable of capturing several fields and creating montage images. It features a 14 megapixel sensor to capture color, red free, and infrared images. The red free photos are extracted from the color image rather than through a separate exposure with a blue-green light source.



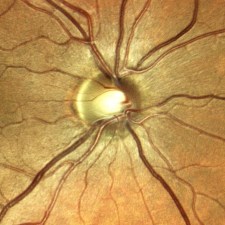 One of the features touted by the manufacturer is that it prevents “optic disc bleaching” seen with some fundus cameras. It does hold detail in optic disc photos, but the flip side to this is that the rim of the nerve can appear abnormally dark or gray, making it difficult to document pallor. Disc bleaching shouldn’t be a problem in fundus cameras that are
One of the features touted by the manufacturer is that it prevents “optic disc bleaching” seen with some fundus cameras. It does hold detail in optic disc photos, but the flip side to this is that the rim of the nerve can appear abnormally dark or gray, making it difficult to document pallor. Disc bleaching shouldn’t be a problem in fundus cameras that are 


























 On a recent trip through Virginia, I stumbled across a gem of a museum tucked away in historic downtown Staunton, VA. I had been browsing brochures of local attractions on a stand in the lobby of my hotel and spotted a photo of a vintage view camera. The brochure was for the Camera Heritage Museum. The non-profit museum bills itself as the largest camera museum on the East Coast. It wasn’t far from my hotel, so I decided to visit, not knowing what to expect.
On a recent trip through Virginia, I stumbled across a gem of a museum tucked away in historic downtown Staunton, VA. I had been browsing brochures of local attractions on a stand in the lobby of my hotel and spotted a photo of a vintage view camera. The brochure was for the Camera Heritage Museum. The non-profit museum bills itself as the largest camera museum on the East Coast. It wasn’t far from my hotel, so I decided to visit, not knowing what to expect.




